Conversions You Need to Know for Chemistry
Learning Objectives
Past the end of this section, you will exist able to:
- Explain the dimensional analysis (gene characterization) approach to mathematical calculations involving quantities
- Use dimensional analysis to carry out unit of measurement conversions for a given property and computations involving two or more properties
It is often the example that a quantity of interest may not be like shooting fish in a barrel (or even possible) to measure directly but instead must be calculated from other directly measured properties and appropriate mathematical relationships. For example, consider measuring the average speed of an athlete running sprints. This is typically achieved by measuring the time required for the athlete to run from the starting line to the stop line, and the altitude between these two lines, so calculating speed from the equation that relates these iii backdrop:
[latex]\text{speed}=\frac{\text{altitude}}{\text{fourth dimension}}[/latex]
An Olympic-quality sprinter tin run 100 m in approximately 10 south, corresponding to an average speed of [latex]\frac{\text{100 chiliad}}{\text{ten s}}=\text{10 m/due south}[/latex].
Note that this simple arithmetic involves dividing the numbers of each measured quantity to yield the number of the computed quantity (100/x = 10) and also dividing the units of each measured quantity to yield the unit of the computed quantity (1000/due south = m/s). At present, consider using this aforementioned relation to predict the time required for a person running at this speed to travel a distance of 25 m. The same relation between the three backdrop is used, but in this case, the two quantities provided are a speed (10 thousand/southward) and a altitude (25 m). To yield the sought property, time, the equation must be rearranged accordingly:
[latex]\text{time}=\frac{\text{distance}}{\text{speed}}[/latex]
The fourth dimension tin and so be computed equally [latex]\frac{\text{25 m}}{\text{10 thousand/s}}=\text{two.5 southward}[/latex].
Again, arithmetic on the numbers (25/x = 2.5) was accompanied by the same arithmetic on the units (one thousand/one thousand/s = s) to yield the number and unit of measurement of the result, 2.5 s. Note that, just as for numbers, when a unit is divided by an identical unit (in this case, thou/one thousand), the result is "i"—or, every bit commonly phrased, the units "abolish."
These calculations are examples of a versatile mathematical approach known as dimensional analysis (or the factor-label method). Dimensional analysis is based on this premise: the units of quantities must be subjected to the same mathematical operations every bit their associated numbers. This method can be applied to computations ranging from simple unit conversions to more complex, multi-step calculations involving several unlike quantities.
Conversion Factors and Dimensional Assay
A ratio of two equivalent quantities expressed with different measurement units can be used as a unit conversion cistron. For example, the lengths of two.54 cm and i in. are equivalent (past definition), and so a unit conversion factor may be derived from the ratio,
[latex]\frac{\text{2.54 cm}}{\text{1 in.}}\text{(2.54 cm}=\text{1 in.) or two.54}\frac{\text{cm}}{\text{in.}}[/latex]
Several other unremarkably used conversion factors are given in Table 1.
| Tabular array ane. Common Conversion Factors | ||
|---|---|---|
| Length | Book | Mass |
| 1 thousand = i.0936 yd | 1 Fifty = i.0567 qt | i kg = 2.2046 lb |
| one in. = two.54 cm (exact) | i qt = 0.94635 L | ane lb = 453.59 g |
| 1 km = 0.62137 mi | 1 ft3 = 28.317 L | 1 (avoirdupois) oz = 28.349 g |
| 1 mi = 1609.three m | 1 tbsp = 14.787 mL | 1 (troy) oz = 31.103 g |
When nosotros multiply a quantity (such as distance given in inches) by an appropriate unit conversion cistron, we convert the quantity to an equivalent value with different units (such as altitude in centimeters). For example, a basketball game histrion's vertical bound of 34 inches tin can be converted to centimeters by:
[latex]\text{34 in.}\times \frac{\text{ii.54 cm}}{ane\cancel{\text{in.}}}=\text{86 cm}[/latex]
Since this simple arithmetic involves quantities, the premise of dimensional analysis requires that we multiply both numbers and units. The numbers of these two quantities are multiplied to yield the number of the production quantity, 86, whereas the units are multiplied to yield [latex]\frac{\text{in.}\times \text{cm}}{\text{in.}}[/latex] . Merely every bit for numbers, a ratio of identical units is also numerically equal to one, [latex]\frac{\text{in.}}{\text{in.}}=\text{one,}[/latex] and the unit production thus simplifies to cm. (When identical units divide to yield a cistron of 1, they are said to "cancel.") Using dimensional analysis, nosotros tin can determine that a unit conversion factor has been set up correctly by checking to ostend that the original unit will cancel, and the result will incorporate the sought (converted) unit.
Example 1: Using a Unit Conversion Factor
The mass of a competition frisbee is 125 g. Catechumen its mass to ounces using the unit of measurement conversion gene derived from the relationship 1 oz = 28.349 g (Table 1).
Check Your Learning
Convert a volume of 9.345 qt to liters.
Across simple unit conversions, the factor-label method can exist used to solve more complex issues involving computations. Regardless of the details, the basic approach is the aforementioned—all the factors involved in the calculation must be appropriately oriented to insure that their labels (units) will appropriately cancel and/or combine to yield the desired unit in the event. This is why it is referred to as the factor-characterization method. Equally your study of chemical science continues, y'all volition encounter many opportunities to apply this approach.
Instance 2: Computing Quantities from Measurement Results and Known Mathematical Relations
What is the density of common antifreeze in units of k/mL? A 4.00-qt sample of the antifreeze weighs ix.26 lb.
Check Your Learning
What is the volume in liters of 1.000 oz, given that ane 50 = 1.0567 qt and 1 qt = 32 oz (exactly)?
Bear witness Answer
[latex]ii.956\times {ten}^{-2}\text{L}[/latex]
Example 3: Computing Quantities from Measurement Results and Known Mathematical Relations
While being driven from Philadelphia to Atlanta, a distance of about 1250 km, a 2014 Lamborghini Aventador Roadster uses 213 L gasoline.
- What (average) fuel economy, in miles per gallon, did the Roadster get during this trip?
- If gasoline costs $3.fourscore per gallon, what was the fuel price for this trip?
Check Your Learning
A Toyota Prius Hybrid uses 59.7 Fifty gasoline to bulldoze from San Francisco to Seattle, a altitude of 1300 km (two significant digits).
- What (average) fuel economy, in miles per gallon, did the Prius get during this trip?
- If gasoline costs $3.90 per gallon, what was the fuel cost for this trip?
Exercises
Book
- The label on a soft drink bottle gives the volume in ii units: two.0 L and 67.6 fl oz. Utilise this information to derive a conversion factor between the English language and metric units. How many significant figures tin can you justify in your conversion factor?
- The label on a box of cereal gives the mass of cereal in two units: 978 grams and 34.5 oz. Use this data to discover a conversion factor betwixt the English and metric units. How many significant figures tin can you justify in your conversion gene?
- How many milliliters of a soft drink are contained in a 12.0-oz tin can?
- A butt of oil is exactly 42 gal. How many liters of oil are in a barrel?
- Many medical laboratory tests are run using 5.0 μL claret serum. What is this volume in milliliters?
- Milk is sold by the liter in many countries. What is the volume of exactly 1/2 gal of milk in liters?
- Gasoline is sold by the liter in many countries. How many liters are required to fill a 12.0-gal gas tank?
- Calculate these volumes.
- What is the volume of 11.iii g graphite, density = 2.25 chiliad/cm3?
- What is the volume of 39.657 g bromine, density = 2.928 g/cm3?
- Calculate these volumes.
- What is the volume of 25 g iodine, density = 4.93 one thousand/cm3?
- What is the volume of 3.28 g gaseous hydrogen, density = 0.089 k/L?
Mass
- Is a 197-lb weight lifter light enough to compete in a class express to those weighing xc kg or less?
- A very adept 197-lb weight lifter lifted 192 kg in a move called the clean and jerk. What was the mass of the weight lifted in pounds?
- If an aspirin tablet contains 325 mg aspirin, how many grams of aspirin does information technology comprise?
- A long ton is defined as exactly 2240 lb. What is this mass in kilograms?
- The gas tank of a certain luxury automobile holds 22.iii gallons according to the owner'southward manual. If the density of gasoline is 0.8206 g/mL, determine the mass in kilograms and pounds of the fuel in a full tank.
- Summate these masses.
- What is the mass of half-dozen.00 cm3 of mercury, density = 13.5939 chiliad/cmthree?
- What is the mass of 25.0 mL octane, density = 0.702 thousand/cm3?
- Calculate these masses.
- What is the mass of 4.00 cm3 of sodium, density = 0.97 g/cm?
- What is the mass of 125 mL gaseous chlorine, density = 3.16 g/L?
Length
- The diameter of a red claret cell is almost 3 × x−iv in. What is its diameter in centimeters?
- The altitude between the centers of the two oxygen atoms in an oxygen molecule is 1.21 × 10−8 cm. What is this altitude in inches?
- A chemist'southward 50-Trillion Angstrom Run (see Exercise 22) would exist an archeologist's 10,900 cubit run. How long is one cubit in meters and in feet? (i Å = ane × 10−eight cm)
- Many chemistry conferences accept held a 50-Trillion Angstrom Run (two significant figures). How long is this run in kilometers and in miles? (ane Å = 1 × 10−10 chiliad)
Exercises
- Write conversion factors (as ratios) for the number of:
- yards in 1 meter
- liters in 1 liquid quart
- pounds in one kilogram
- Write conversion factors (equally ratios) for the number of:
- kilometers in i mile
- liters in 1 cubic pes
- grams in 1 ounce
- Soccer is played with a circular ball having a circumference betwixt 27 and 28 in. and a weight between 14 and 16 oz. What are these specifications in units of centimeters and grams?
- A woman's basketball has a circumference between 28.5 and 29.0 inches and a maximum weight of 20 ounces (two significant figures). What are these specifications in units of centimeters and grams?
- Use scientific (exponential) note to express the post-obit quantities in terms of the SI base of operations units in Table one:
- 0.xiii g
- 232 Gg
- 5.23 pm
- 86.3 mg
- 37.six cm
- 54 μm
- one Ts
- 27 ps
- 0.xv mK
- Complete the following conversions betwixt SI units.
- 612 g = ________ mg
- 8.160 m = ________ cm
- 3779 μg = ________ g
- 781 mL = ________ L
- 4.18 kg = ________ 1000
- 27.8 grand = ________ km
- 0.xiii mL = ________ L
- 1738 km = ________ yard
- 1.nine Gg = ________ g
- Make the conversion indicated in each of the following:
- the men's world tape long jump, 29 ft iv¼ in., to meters
- the greatest depth of the body of water, about vi.5 mi, to kilometers
- the area of the country of Oregon, 96,981 mi2, to square kilometers
- the volume of 1 gill (exactly 4 oz) to milliliters
- the estimated volume of the oceans, 330,000,000 mi3, to cubic kilometers.
- the mass of a 3525-lb machine to kilograms
- the mass of a 2.3-oz egg to grams
- Make the conversion indicated in each of the post-obit:
- the length of a soccer field, 120 chiliad (three significant figures), to anxiety
- the height of Mt. Kilimanjaro, at 19,565 ft the highest mountain in Africa, to kilometers
- the area of an 8.v t 11-inch sheet of paper in cm2
- the displacement volume of an automobile engine, 161 in.iii, to liters
- the estimated mass of the atmosphere, 5.six t 10xv tons, to kilograms
- the mass of a bushel of rye, 32.0 lb, to kilograms
- the mass of a 5.00-grain aspirin tablet to milligrams (1 grain = 0.00229 oz)
- Equally an instructor is preparing for an experiment, he requires 225 g phosphoric acid. The only container readily available is a 150-mL Erlenmeyer flask. Is it large enough to comprise the acid, whose density is 1.83 thousand/mL?
- To set for a laboratory period, a student lab assistant needs 125 g of a compound. A bottle containing ane/four lb is bachelor. Did the student have enough of the chemical compound?
- 27 A chemistry pupil is 159 cm tall and weighs 45.8 kg. What is her pinnacle in inches and weight in pounds?
- In a recent Chiliad Prix, the winner completed the race with an average speed of 229.eight km/h. What was his speed in miles per hr, meters per second, and feet per second?
- Solve these problems about lumber dimensions.
- To describe to a European how houses are constructed in the US, the dimensions of "ii-past-iv" lumber must exist converted into metric units. The thickness × width × length dimensions are ane.50 in. × 3.fifty in. × viii.00 ft in the US. What are the dimensions in cm × cm × m?
- This lumber can be used every bit vertical studs, which are typically placed xvi.0 in. apart. What is that altitude in centimeters?
- Summate the density of aluminum if 27.half-dozen cmiii has a mass of 74.half-dozen k.
- Osmium is one of the densest elements known. What is its density if ii.72 m has a volume of 0.121 cmiii?
Conversion of Temperature Units
Nosotros use the word temperature to refer to the hotness or coldness of a substance. One way we measure a change in temperature is to apply the fact that most substances expand when their temperature increases and contract when their temperature decreases. The mercury or alcohol in a mutual drinking glass thermometer changes its volume as the temperature changes. Because the book of the liquid changes more than the book of the glass, nosotros can see the liquid expand when information technology gets warmer and contract when it gets cooler.
To mark a calibration on a thermometer, we need a ready of reference values: Two of the nearly ordinarily used are the freezing and boiling temperatures of water at a specified atmospheric pressure. On the Celsius scale, 0 °C is divers as the freezing temperature of water and 100 °C equally the humid temperature of water. The space between the two temperatures is divided into 100 equal intervals, which nosotros phone call degrees. On the Fahrenheit calibration, the freezing point of water is defined every bit 32 °F and the boiling temperature as 212 °F. The space between these two points on a Fahrenheit thermometer is divided into 180 equal parts (degrees).
Defining the Celsius and Fahrenheit temperature scales as described in the previous paragraph results in a slightly more complex relationship between temperature values on these 2 scales than for unlike units of measure for other properties. Most measurement units for a given belongings are direct proportional to ane another (y = mx). Using familiar length units as one instance:
[latex]\text{length in feet}=\left(\frac{\text{i ft}}{\text{12 in.}}\right)\times \text{length in inches}[/latex]
where y = length in feet, ten = length in inches, and the proportionality constant, m, is the conversion gene. The Celsius and Fahrenheit temperature scales, all the same, practise not share a common nothing signal, so the relationship between these two scales is a linear one rather than a proportional one (y = mx + b). Consequently, converting a temperature from one of these scales into the other requires more than simple multiplication past a conversion gene, one thousand, it also must accept into business relationship differences in the scales' zero points (b).
The linear equation relating Celsius and Fahrenheit temperatures is easily derived from the two temperatures used to define each calibration. Representing the Celsius temperature as x and the Fahrenheit temperature equally y, the slope, 1000, is computed to be:
[latex]\displaystyle{m}=\frac{\Delta y}{\Delta 10}=\frac{212^{\circ}\text{ F}-32^{\circ}\text{ F}}{100^{\circ}\text{ C}-0^{\circ}\text{ C}}=\frac{180^{\circ}\text{ F}}{100^{\circ}\text{ C}}=\frac{ix^{\circ}\text{ F}}{five^{\circ}\text{ C}}[/latex]
The y-intercept of the equation, b, is then calculated using either of the equivalent temperature pairs, (100 °C, 212 °F) or (0 °C, 32 °F), equally:
[latex]b=y-mx=32^{\circ}\text{ F}-\frac{9 ^{\circ}\text{ F}}{5 ^{\circ}\text{ C}}\times{0}^{\circ}\text{ C}=32^{\circ}\text{ F}[/latex]
The equation relating the temperature scales is so:
[latex]{T}_{\text{ }^{\circ}\text{F}}=\left(\frac{nine^{\circ}\text{ F}}{5^{\circ}\text{ C}}\times{T}_{\text{ }^{\circ}\text{C}}\right)+32 ^{\circ}\text{ C}[/latex]
An abbreviated form of this equation that omits the measurement units is:
[latex]{T}_{\text{ }^{\circ}\text{F}}=\frac{nine}{5}\times{T}_{^{\circ}\text{ C}}+32[/latex]
Rearrangement of this equation yields the form useful for converting from Fahrenheit to Celsius:
[latex]{T}_{\text{ }^{\circ}\text{C}}=\frac{5}{nine}\left({T}_{^{\circ}\text{ F}}-32\right)[/latex]
Every bit mentioned earlier in this chapter, the SI unit of temperature is the kelvin (Thou). Unlike the Celsius and Fahrenheit scales, the kelvin scale is an absolute temperature calibration in which 0 (zero) K corresponds to the lowest temperature that tin can theoretically be achieved. The early 19th-century discovery of the relationship between a gas's volume and temperature suggested that the volume of a gas would be zero at −273.fifteen °C. In 1848, British physicist William Thompson, who subsequently adopted the title of Lord Kelvin, proposed an accented temperature scale based on this concept (further treatment of this topic is provided in this text'south chapter on gases).
The freezing temperature of water on this scale is 273.fifteen Yard and its humid temperature 373.fifteen K. Observe the numerical difference in these 2 reference temperatures is 100, the aforementioned equally for the Celsius scale, and and then the linear relation between these two temperature scales will exhibit a slope of [latex]1\frac{\text{Chiliad}}{^{\circ}\text{C}}[/latex] . Following the aforementioned arroyo, the equations for converting between the kelvin and Celsius temperature scales are derived to be:
[latex]{T}_{\text{Thou}}={T}_{^{\circ}\text{C}}+\text{273.fifteen}[/latex]
[latex]{T}_{\text{ }^{\circ}\text{C}}={T}_{\text{Thou}}-\text{273.15}[/latex]
The 273.15 in these equations has been determined experimentally, so it is not exact. Figure 1 shows the relationship amid the iii temperature scales. Remember that we do not use the caste sign with temperatures on the kelvin scale.
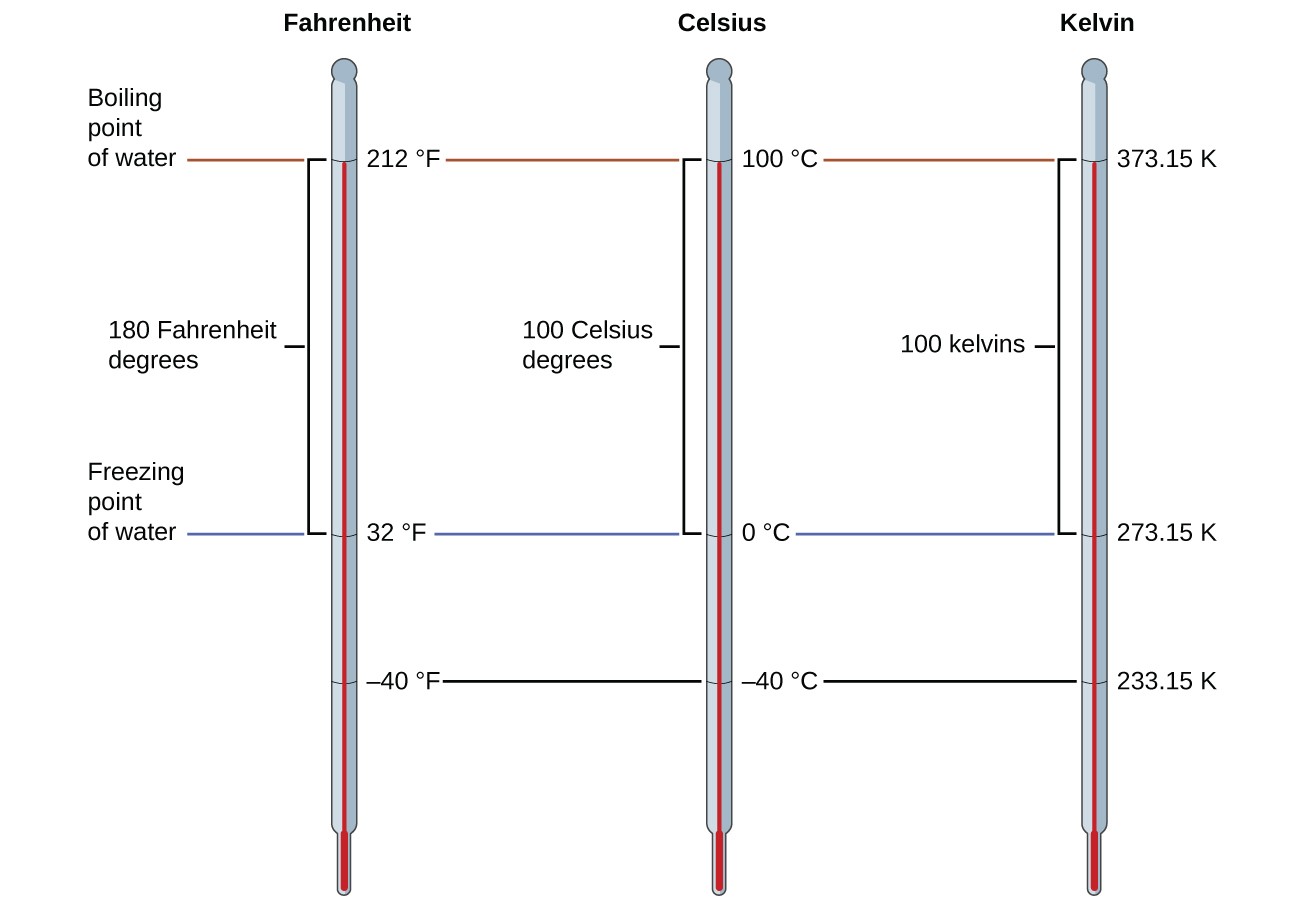
Effigy 1. The Fahrenheit, Celsius, and kelvin temperature scales are compared.
Although the kelvin (absolute) temperature scale is the official SI temperature scale, Celsius is commonly used in many scientific contexts and is the scale of pick for nonscience contexts in most all areas of the world. Very few countries (the U.S. and its territories, the Bahamas, Belize, Cayman Islands, and Palau) still utilize Fahrenheit for weather, medicine, and cooking.
Example iv:Conversion from Celsius
Normal torso temperature has been commonly accepted equally 37.0 °C (although information technology varies depending on time of 24-hour interval and method of measurement, as well as among individuals). What is this temperature on the kelvin calibration and on the Fahrenheit calibration?
Check Your Learning
Convert eighty.92 °C to Yard and °F.
Testify Reply
354.07 M, 177.7 °F
Example 5:Conversion from Fahrenheit
Baking a ready-fabricated pizza calls for an oven temperature of 450 °F. If y'all are in Europe, and your oven thermometer uses the Celsius scale, what is the setting? What is the kelvin temperature?
Check Your Learning
Catechumen fifty °F to °C and K.
Central Concepts and Summary
Measurements are made using a variety of units. It is ofttimes useful or necessary to catechumen a measured quantity from ane unit of measurement into another. These conversions are achieved using unit conversion factors, which are derived past simple applications of a mathematical approach called the factor-label method or dimensional assay. This strategy is also employed to calculate sought quantities using measured quantities and appropriate mathematical relations.
Fundamental Equations
- [latex]{T}_{^{\circ}\text{C}}=\frac{5}{nine}\times {T}_{^{\circ}\text{F}}-32[/latex]
- [latex]{T}_{^{\circ}\text{F}}=\frac{9}{5}\times {T}_{^{\circ}\text{C}}+32[/latex]
- [latex]{T}_{\text{1000}}=\text{ }^{\circ}\text{C}+273.15[/latex]
- [latex]{T}_{^{\circ}\text{C}}=\text{Grand}-273.15[/latex]
Exercises
- Convert the humid temperature of gold, 2966 °C, into degrees Fahrenheit and kelvin.
- Catechumen the temperature of scalding water, 54 °C, into degrees Fahrenheit and kelvin.
- Convert the temperature of the coldest expanse in a freezer, −ten °F, to degrees Celsius and kelvin.
- Convert the temperature of dry ice, −77 °C, into degrees Fahrenheit and kelvin.
- Convert the humid temperature of liquid ammonia, −28.i °F, into degrees Celsius and kelvin.
- The label on a pressurized tin can of spray disinfectant warns against heating the can higher up 130 °F. What are the corresponding temperatures on the Celsius and kelvin temperature scales?
- The weather in Europe was unusually warm during the summer of 1995. The Idiot box news reported temperatures as high as 45 °C. What was the temperature on the Fahrenheit scale?
Glossary
dimensional assay: (besides, factor-characterization method) versatile mathematical approach that tin be applied to computations ranging from simple unit conversions to more complex, multi-step calculations involving several different quantities
Fahrenheit: unit of temperature; water freezes at 32 °F and boils at 212 °F on this scale
unit conversion factor: ratio of equivalent quantities expressed with dissimilar units; used to convert from one unit to a different unit
Source: https://courses.lumenlearning.com/introductorychemistry/chapter/mathematical-treatment-of-measurement-results/
0 Response to "Conversions You Need to Know for Chemistry"
Post a Comment